When you find out that a medicine is not available in your country, it can feel like a dead end or the end of the road.
In many cases, it isn’t.
If a treatment has been approved in another country or is still being developed, patients might be able to access it through legal channels before it becomes available locally.
These channels are valid ways for patients to obtain medicines that are not yet available locally.
This guide explains what early access means, the legal options available, and what patients should know before exploring cross-border access to medicine.
What Is Early Access to Medicines?
“Early access” refers to regulated programs that allow patients to receive medicines outside the standard commercial supply chain when:
- The medicine is approved in another country but not yet available locally.
- The medicine is still under review by regulators.
- The patient has a serious or life-threatening condition with few treatment options.
Different countries use different terms, but common mechanisms include:
- Expanded Access Programs (EAP)
- Compassionate Use Programs
- Named Patient Importation Pathways
These programs are designed for patients who cannot wait for routine market availability.
Who Early Access Programs Are Typically For
Rare disease: A patient with a rare genetic condition might find out that a targeted therapy is available in another region, but not yet provided locally. If there are no effective alternatives, a physician may explore regulated methods to request access for that specific patient.
Oncology: A patient with advanced cancer may have a tumor biomarker that aligns with a newly approved targeted therapy in the US or EU, but the medicine is not available through local hospitals yet. In some cases, a doctor might consider early access options while ensuring the process complies with national regulations.
Life-threatening illness: For rapidly progressive or life-threatening conditions where waiting could significantly worsen outcomes, doctors sometimes assess early access options when standard treatments have failed or are not appropriate, and the benefit-risk profile supports access.
When time is critical, the question is not only what exists globally, but what is legally possible for a specific patient right now under medical supervision.
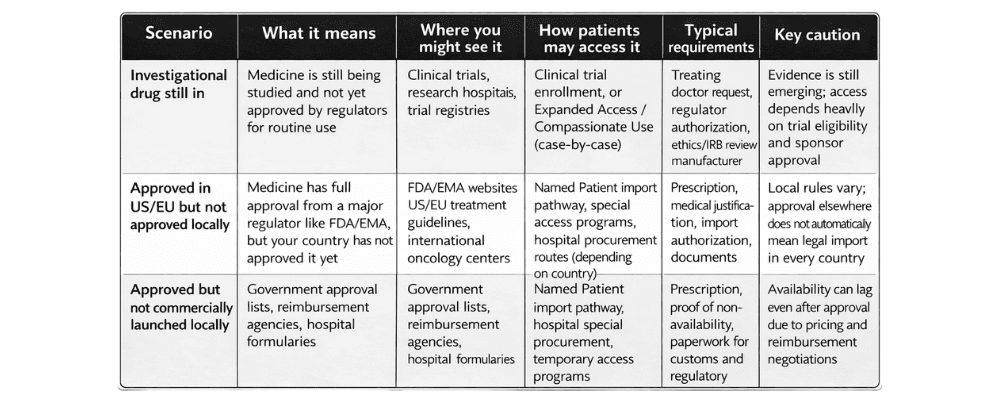
Expanded Access (United States)
In the United States, the FDA regulates Expanded Access, sometimes known as compassionate use, through formal rules. These rules allow patients with serious diseases to receive investigational drugs outside clinical trials.
Key points:
- The treating physician must determine that no comparable alternative exists
- The potential benefit must justify the potential risks
- The manufacturer must agree to provide the drug
- The FDA must authorize access
- An Institutional Review Board (IRB) must review the request
Compassionate Use (European Union)
In the European Union, compassionate use programs provide access to unapproved medicines for patients with chronic, serious, or life-threatening diseases when no satisfactory authorized therapies are available.
These programs are coordinated at the national level but guided by the European Medicines Agency (EMA).
Each EU country implements its own procedures under this framework.
Named Patient Importation Pathways
Many countries let physicians request unapproved or non-locally marketed medicines for individual patients through what is often called a Named Patient pathway.
While rules vary by country, common elements include:
- A valid prescription from a licensed physician
- Confirmation that the medicine is not available locally
- Documentation stating the medicine is for personal use
- Regulatory forms required by the importing country
- Compliance with customs and pharmaceutical import laws
These systems are used by hospitals and doctors worldwide and are regulated by national health authorities.
Myth vs Fact: Understanding Legal Medicine Import
Myth: Importing medicine is a black market activity
Fact: Legal early access pathways exist in many countries. When done through proper regulatory channels, with physician oversight and in compliance with documentation requirements, importing medicine can be legal and regulated.
Myth: You must wait for local approval
Fact: In many areas, access may be possible once a medicine is approved by a recognized regulatory authority, such as the FDA or EMA, even if local approval is still pending. The specific eligibility depends on national laws.
Myth: A prescription is all you need
Fact: A prescription is usually only the starting point. Additional documentation often includes:
- Patient declaration forms
- Regulatory applications
- Import authorization paperwork
- Customs documentation
- In some cases, physician justification letters
Missing paperwork can result in delays or rejection.
Why Timing Matters
It can take several years for a newly approved medicine to become available for sale in every country. Regulatory review timelines, pricing negotiations, and reimbursement processes can vary widely.
While regulatory systems work carefully to ensure safety and quality, serious illness may leave patients waiting years for treatment.
However, any decision to pursue early access should be:
- Based on medical advice
- Evaluated for risk vs benefit
- Conducted strictly within legal frameworks
Waiting should be a medical decision, not just a bureaucratic outcome.
Example timeline :
A cancer medicine approved by the FDA in 2024 may still take 2 to 5 years to become widely available in another country due to additional steps, including local regulatory review, pricing negotiations, reimbursement decisions, and supply chain onboarding.
During this gap, some patients may qualify for regulated early access options, depending on local rules.
What Patients Should Do First
If you are told a treatment is not available locally:
- Confirm whether the medicine is approved elsewhere
- Ask your doctor whether early access programs apply
- Verify whether your country has a Named Patient or similar pathway
- Understand documentation requirements
- Confirm the source pharmacy or supplier complies with export laws
Patients should never purchase prescription medicines from unverified online sources that claim to bypass regulations.
The World Health Organization warns about the risks posed by falsified and substandard medicines in unregulated markets.
Compliance and Safety Considerations
Legal early access should always include:
- Physician supervision
- Verified manufacturer sourcing
- Temperature-controlled shipping when required
- Proper customs declaration
- Regulatory documentation
- Clear pharmacovigilance tracking
Cross-border access to medicine must comply with the pharmaceutical regulations of both exporting and importing countries.
A Balanced Perspective
Early access pathways exist to support patients with serious conditions who have limited options. They are structured, regulated, and legally recognized mechanisms.
However:
- Not every medicine qualifies
- Not every country allows imports in the same way
- Not every patient meets eligibility criteria
- Manufacturer participation may be required
Patients deserve transparency about both possibilities and limitations.
Final Thought
“Not available locally” does not always mean “does not exist.” It may mean approval has not yet arrived in your country.
Legal early access pathways like Expanded Access, Compassionate Use, and Named Patient programs exist to bridge that gap. When used properly under physician guidance and in full compliance with regulatory requirements, they can be beneficial.
If you are considering this route, begin with a medical discussion. Verify your country’s regulations, and ensure every step follows the correct legal procedures.
Health decisions should be informed, compliant, and guided by professionals, especially when they involve medicines across borders.
References
US Food and Drug Administration. Expanded Access.
European Medicines Agency. Compassionate use.
UK Medicines and Healthcare products Regulatory Agency. Supplying unlicensed medicinal products (Specials).
World Health Organization. Substandard and falsified medical products.






